![]()
An overview of available Hypoglycemic Triterpenoids and Saponins to cure Diabetes mellitus
Dadu Khan Burdi1, Sumera Qureshi1*, Allah Bux Ghanghro2
Adv. life sci., vol. 1, no. 3, pp. 119-128, May 2014
*Corresponding Author: Sumera Qureshi (Email: s.dr582@gmail.com)
Author Affiliations
2- Institute of Biochemistry, University of Sindh, Karachi – Pakistan
Abstract![]()
Introduction
Methods
Discussion
Conclusion
References
Abstract
Diabetes mellitus is a condition when excess amount of sugar is excreted out in the urine. It is probably caused due to hyperglycemia; when body fails to produce sufficient amount of insulin which may utilize or help to store these excessive carbohydrates. When a body does not produce sufficient insulin or to help utilize carbohydrates, it results in the accumulation of unutilized sugar in the blood, the condition is termed as hyperglycemia whereas the condition of passing off the excess sugar in the urine is known as diabetes mellitus. The excretion of sugar makes a profound effect on health that may lead to disability and death. According to some studies, it may cause myocardial infarction, cardiovascular disorders and terminal nephritis. These complications are reported to be the most important causes of mortality and the principal cause of irreversible blindness. There are hundreds of millions of patients around the world suffering from this disease and the number is spreading with an alarming rate. Such a condition has inspired the therapists to develop the methods which help in controlling this malaise. In this article, we have summarized some of hypoglycemic agents from natural sources especially from plants. Since there is a vast number of plants, which are reported to be utilized traditionally in the crude form for diabetes cure in the past. This article is meant to mention only the hypoglycemic ingredients of triterpenoids origin. This information can be helpful in getting new and more effective drugs in future by utilizing unexplored plants which are reported in the literature to possess hypoglycemic activity. This current review is comprised of the relevant work done up to the year 2007 based on search from Google.
Keywords: Hyperglycemia, Diabetes mellitus, Hypoglycemic triterpenoids, Saponins
Introduction
There are quite a large number of people who suffer from diabetes that may result from hyperglycemia. According to one estimate there were 124.7 million cases of diabetes mellitus in the recent past around the world [1-3]. Whereas another study reveals that the number is expected to increase by 2025 to 300 million from 151 million as recorded in the year 2000 [2]. With the passage of time there is an exponential growth in the number of patients with diabetes mellitus and it would be a leading cause of higher death rates in the near future. It therefore, has become a big challenge to the therapists to devise the methods to effectively control this disease. There is a need of exploring the effective ways to bring about awareness to the masses regarding prevention measures and proper ways and means to cure the disease, diabetes mellitus. If such efforts are not dealt with properly and seriously, the disease could be a leading cause of deaths. A number of drugs have been discovered in this regard. However, the disease still has a profound effect on human life. Nowadays as the phytochemical research has acquired a predominant position in the field of medicine, the interest is advanced to explore the plants which may prove to be beneficial for the control of this disease.
Methods
Search strategy and selection criteria
A systematic search was carried out from Google by entering keywords like, “Hypoglycemic Triterpenoids” and “saponins’’ with no filter. We found about 36,000 results, from which only 80 relevant articles.
Discussion
Hypoglycemic constituents Role
The compound which brings the excessive sugar level towards the normal range by certain biochemical reactions is known as hypoglycemic component. There are several classes of organic compound which possess hypoglycemic activity such as terpenoids, steroids, saponins flavonoids, alkaloids, glycans proteins [2]. To cite all of them is beyond the scope of this article. Hence the compound of triterpeniod origin obtained from various plants is described. Hence the hypoglycemic compounds of triterpeniod origin reported since the beginning of systematic investigation up to the year 2007 from various plants are described. The relevant constituents are summarized in tables (1- 6) which are specified with the particular groups of triterpenoids.
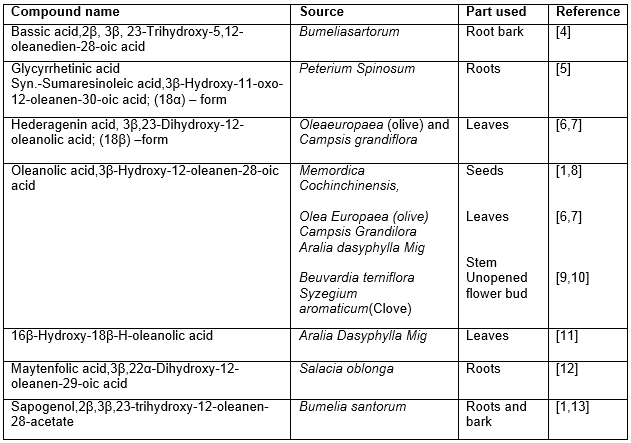
Table 1: Hypoglycemic triterpenoids of oleanane origin
Triterpenoids and saponins with hypoglycemic activity:
Most of the hypoglycemic triterpenoids (Table 1) and their glycosides (Table 2-3) belong to oleanane group. Some other groups with the similar activity comprising of ursane (Table 4), lupane (Table 5) are reported to a lesser extent.
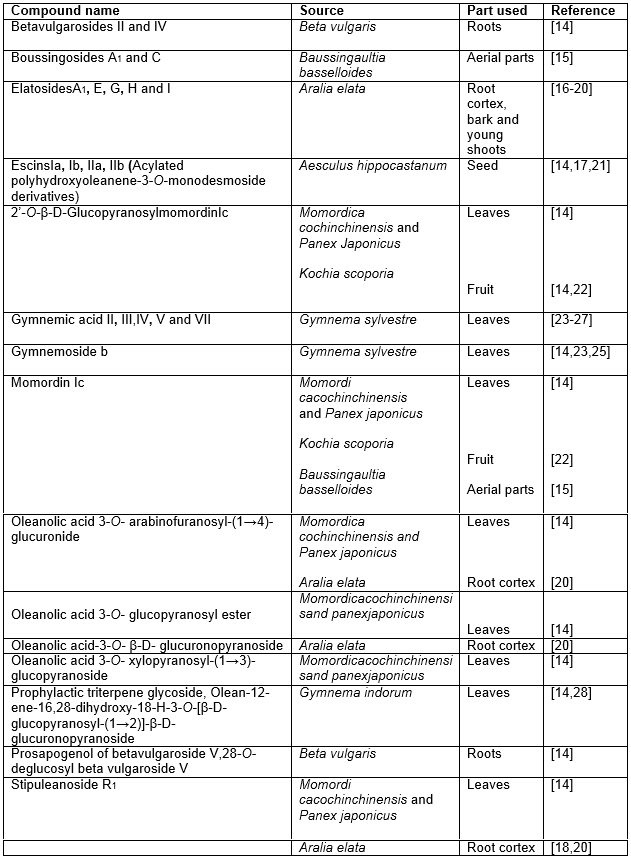
Table 2: Hypoglycemic monodesmosidesaponins of oleanane origin
The plants which are reported to possess hypoglycemic activity may be taken under consideration for being the source of these important bioactive compounds with specific reference to being anti-hyperglycemic activity. In the literature, there are lot of plants which are used traditionally for treating diabetes mellitus [1-3]. Some of them which are mentioned, along with the antidiabetic constituents, in this article are, Olea europea, Campsis grandiflora, Beta vulgaris, Gymnema sylvestre and Panex ginsing.
The isolation of several monodesmoside derivatives (Table 2) and bisdesmoside derivatives (Table 3) of oleanolic acid obtained from various plant species reported to be hypoglycemic are worth noting as these groups of compounds have larger contribution in this respect.
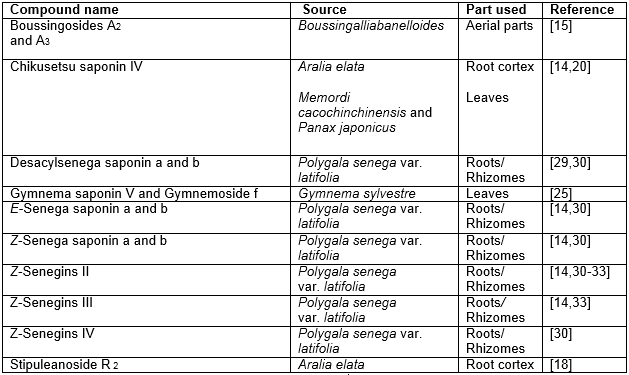
Table 3: Hypoglycemic bisdesmosidesaponins of oleanane origin
Table 4: Hypoglycemic triterpenoids of ursane origin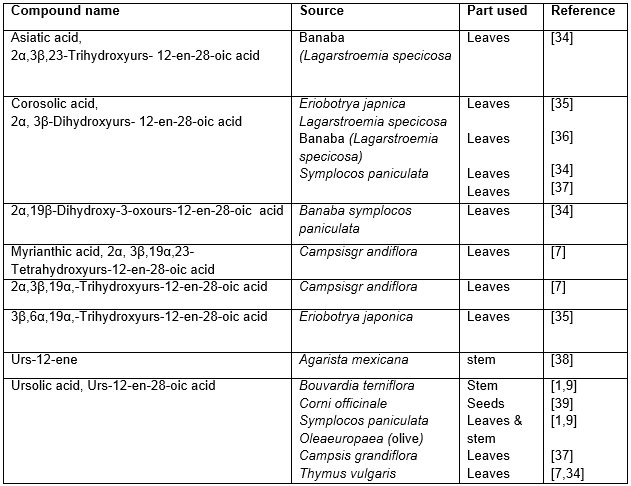
Table 5: Hypoglycemic triterpenoids of lupane origin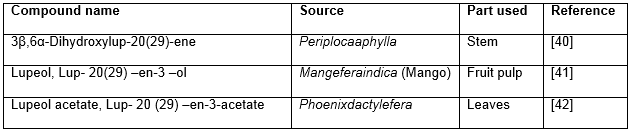
Table 6: Miscellaneous Hypoglycemic triterpenoids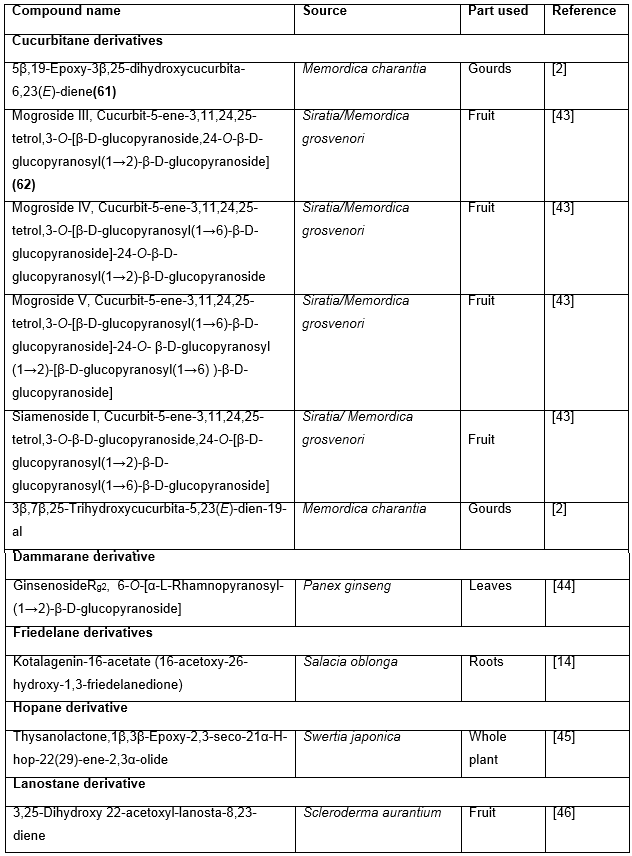
Conclusion
The work by coordinating the activity of a particular plant used for treatment of diabetes, to the presence of antidiabetic agents hitherto reported may lead to very promising results. By viewing above data related to the hypoglycemic activity of the aforementioned compounds, it seems that these ingredients may be exploited for large scale supply of the drugs at industrial scale to cope with the increasing demand of such drugs. The objective can also be achieved by utilizing the constituents available in abundance, which are closely related to the framework of the active principles by bringing them in conformity to the activity positive entity by certain chemical transformations. The promising results may be achieved by exploiting the experiments which have been conducted to check the structure-activity relationship. For example, the sugar moieties attached at position C-3 of the olenane framework enhance the hypoglycemic activity of the relevant glycoside which may be either mono- or oligoglycoside; whereas the attachment of sugar groups at position C-28 decreases or render the molecule biologically inactive [17-20,31,47].
Competing interests: The authors declare no competing interests. Although, the sources of some known compounds may be more than those reported in the article. The incorporation of only selected plants is due to their citation as a source of isolation of respective hypoglycemic constituents.
Acknowledgement. One of the authors (D.K. Burdi) is grateful to Professor V. U. Ahmad for providing him library facilities at International Center for Chemical and Biological Sciences, H.E.J. Research Institute of Chemistry, University of Karachi, Pakistan.
References
- Perez G R, Zavala S M, Perez G S, Perez G C. Antidiabetic effect of compounds isolated from plants. Phytomedicine, (1998); 5(1): 55-75.
- Harinantenaina L, Tanaka M, Takaoka S, Oda M, Mogami O, et al. Momordica charantia constituents and antidiabetic screening of the isolated major compounds. Chemical and Pharmaceutical Bulletin, (2006); 54(7): 1017-1021.
- Zaman K. Medicinal plants with hypoglycemic activity. Journal of Ethnopharmacology, (1989); 26(1): 1-55.
- Naik SR, Dhuley JN, Deshmukh V. Probable mechanism of hypoglycemic activity of bassic acid, a natural product isolated from Bumelia sartorum. Journal of Ethnopharmacology, (1991); 33(1): 37-44.
- Wen X, Sun H, Liu J, Wu G, Zhang L, et al. Pentacyclic triterpenes. Part 1: The first examples of naturally occurring pentacyclic triterpenes as a new class of inhibitors of glycogen phosphorylases. Bioorganic & medicinal chemistry letters, (2005); 15(22): 4944-4948.
- Somova L, Shode F, Ramnanan P, Nadar A. Antihypertensive, antiatherosclerotic and antioxidant activity of triterpenoids isolated from Olea europaea, subspecies africana leaves. Journal of Ethnopharmacology, (2003); 84(2): 299-305.
- Jung S, Ha Y, Shim E, Choi S, Jin J, et al. Insulin-mimetic and insulin-sensitizing activities of a pentacyclic triterpenoid insulin receptor activator. Biochem J, (2007); 403243-250.
- Jalil U, Jabbar A, Hasan C. Hypoglycemic activities of the glycosides of Momordica cochinchinensis. J Bangladesh Acad Sci, (1986); 10(1): 25-30.
- Perez G R, Perez G C, Perez G S, Zavala S M. Effect of triterpenoids of< i> Bouvardia terniflora</i> on blood sugar levels of normal and alloxan diabetic mice. Phytomedicine, (1998); 5(6): 475-478.
- Somova L, Nadar A, Rammanan P, Shode F. Cardiovascular, antihyperlipidemic and antioxidant effects of oleanolic and ursolic acids in experimental hypertension. Phytomedicine, (2003); 10(2): 115-121.
- Yi Y, Gu J, Xiao K, Wang Z, Lin H. [Studies on triterpenoids and their glycosides from Aralia dasyphylla Miq]. Yao xue xue bao= Acta pharmaceutica Sinica, (1997); 32(10): 769-772.
- Lee H-S. Cuminaldehyde: aldose reductase and α-glucosidase inhibitor derived from Cuminum cyminum L. seeds. Journal of agricultural and food chemistry, (2005); 53(7): 2446-2450.
- Nobrega Almeida R, Barbosa Filho M, Ramnath Naik S. Chemistry and pharmacology of an ethanol extract of Bumelia sartorum. Journal of Ethnopharmacology, (1985); 14(2): 173-185.
- Yoshikawa M, Matsuda H. Antidiabetogenic activity of oleanolic acid glycosides from medicinal foodstuffs. Biofactors, (2000); 13(1): 231-237.
- Espada A, Rodriguez J, Villaverde MC, Riguera R. Hypoglucaemic triterpenoid saponins from Boussingaultia baselloides. Canadian journal of chemistry, (1990); 68(11): 2039-2044.
- Clarke MA, Edye LA. Sugar Beet and Sugarcane as Renewable Resources; 1996. ACS Publications. pp. 229-247.
- Yoshikawa M, Harada E, Matsuda H, Murakami T, Yamahara J, et al. Elatosides A and B, potent inhibitors of ethanol absorption in rats from the bark of Aralia elata Seem: the structure-activity relationships of oleanolic acid oligoglycosides. Chemical & pharmaceutical bulletin, (1993); 41(11): 2069-2071.
- Yoshikawa M, Matsuda H, Harada E, Murakami T, Wariishi N, et al. Elatoside E, a new hypoglycemic principle from the root cortex of Aralia elata Seem.: structure-related hypoglycemic activity of oleanolic acid glycosides. Chemical & pharmaceutical bulletin, (1994); 42(6): 1354-1356.
- Yoshikawa M, Yoshizumi S, Ueno T, Matsuda H, Murakami T, et al. Medicinal foodstuffs. I. Hypoglycemic constituents from a garnish foodstuff" taranome," the young shoot of Aralia elata SEEM.: elatosides G, H, I, J, and K. Chemical & pharmaceutical bulletin, (1995); 43(11): 1878-1882.
- Yoshikawa M, Murakami T, Harada E, Murakami N, Yamahara J, et al. Bioactive saponins and glycosides. VI. Elatosides A and B, potent inhibitors of ethanol absorption, from the bark of Aralia elata SEEM.(Araliaceae): the structure-requirement in oleanolic acid glucuronide-saponins for the inhibitory activity. Chemical & pharmaceutical bulletin, (1996); 44(10): 1915-1922.
- Matsuda H, Li Y, Murakami T, Ninomiya K, Yamahara J, et al. Effects of escins Ia, Ib, IIa, and IIb from horse chestnut, the seeds of Aesculus hippocastanum L., on acute inflammation in animals. Biological & pharmaceutical bulletin, (1997); 20(10): 1092-1095.
- Yoshikawa M, Shimada H, Morikawa T, Yoshizumi S, Matsumura N, et al. Medicinal foodstuffs. VII. On the saponin constituents with glucose and alcohol absorption-inhibitory activity from a food garnish" Tonburi", the fruit of Japanese Kochia scoparia (L.) Schrad.: structures of scoparianosides A, B, and C. Chemical & pharmaceutical bulletin, (1997); 45(8): 1300-1305.
- Murakami N, Murakami T, Kadoya M, Matsuda H, Yamahara J, et al. New hypoglycemic constituents in" gymnemic acid" from Gymnema sylvestre. Chemical & pharmaceutical bulletin, (1996); 44(2): 469-471.
- Yoshikawa M, Murakami T, Matsuda H. Medicinal foodstuffs. X. Structures of new triterpene glycosides, gymnemosides-c,-d,-e, and-f, from the leaves of Gymnema sylvestre R. Br.: influence of gymnema glycosides on glucose uptake in rat small intestinal fragments. Chemical and Pharmaceutical Bulletin, (1997); 45(12): 2034-2038.
- Yoshikawa M, Murakami T, Kadoya M, Li Y, Murakami N, et al. Medicinal foodstuffs. IX. The inhibitors of glucose absorption from the leaves of Gymnema sylvestre R. BR.(Asclepiadaceae): structures of gymnemosides a and b. Chemical & pharmaceutical bulletin, (1997); 45(10): 1671-1676.
- Nakamura Y, Tsumura Y, Tonogai Y, Shibata T. Fecal steroid excretion is increased in rats by oral administration of gymnemic acids contained in Gymnema sylvestre leaves. The Journal of nutrition, (1999); 129(6): 1214-1222.
- Sugihara Y, Nojima H, Matsuda H, Murakami T, Yoshikawa M, et al. Antihyperglycemic effects of gymnemic acid IV, a compound derived from Gymnema sylvestre leaves in streptozotocin-diabetic mice. Journal of Asian natural products research, (2000); 2(4): 321-327.
- Yadav M, Lavania A, Tomar R, Prasad G, Jain S, et al. Complementary and comparative study on hypoglycemic and antihyperglycemic activity of various extracts of Eugenia jambolana seed, Momordica charantia fruits, Gymnema sylvestre, and Trigonella foenum graecum seeds in rats. Applied biochemistry and biotechnology, (2010); 160(8): 2388-2400.
- Loew D, Schrödter A, Schwankl W, März R. Measurement of the bioavailability of aescin-containing extracts. Methods Find Exp Clin Pharmacol, (2000); 22(7): 537.
- Yoshikawa M, Murakami T, Ueno T, Kadoya M, Matsuda H, et al. Bioactive saponins and glycosides. I. Senegae radix.(1): E-senegasaponins a and b and Z-senegasaponins a and b, their inhibitory effect on alcohol absorption and hypoglycemic activity. Chemical & pharmaceutical bulletin, (1995); 43(12): 2115-2122.
- Lacaille-Dubois M-A, Mitaine-Offer A-C. Triterpene saponins from Polygalaceae. Phytochemistry Reviews, (2005); 4(2-3): 139-149.
- Kako M, Miura T, Nishiyama Y, Ichimaru M, Moriyasu M, et al. Hypoglycemic effect of the rhizomes of Polygala senega in normal and diabetic mice and its main component, the triterpenoid glycoside senegin-II. Planta medica, (1996); 62(05): 440-443.
- Kako M, Miura T, Nishiyama Y, Ichimaru M, Moriyasu M, et al. Hypoglycemic activity of some triterpenoid glycosides. Journal of natural products, (1997); 60(6): 604-605.
- Matsuyama F, Seino Y, Yamada Y, Hosokawa M, Fujimoto S, et al. Corosolic acid and its analogs as oral gluconeogenesis inhibiting agents. US patent WO2005-JP8569, (2005).
- De Tommasi N, De Simone F, Cirino G, Cicala C, Pizza C. Hypoglycemic effects of sesquiterpene glycosides and polyhydroxylated triterpenoids of Eriobotrya japonica. Planta medica, (1991); 57(05): 414-416.
- Murakami C, Myoga K, Kasai R, Ohtani K, Kurokawa T, et al. Screening of plant constituents for effect on glucose transport activity in Ehrlich ascites tumour cells. Chemical & pharmaceutical bulletin, (1993); 41(12): 2129-2131.
- Na M, Yang S, He L, Oh H, Kim BS, et al. Inhibition of protein tyrosine phosphatase 1B by ursane-type triterpenes isolated from Symplocos paniculata. Planta medica, (2006); 72(3): 261.
- Perez G, Vargas S. Triterpenes from Agarista mexicana as potential antidiabetic agents. Phytotherapy research, (2002); 16(1): 55-58.
- Yamahara J, Mibu H, Sawada T, Fujimura H, Takino S, et al. [Biologically active principles of crude drugs. Antidiabetic principles of corni fructus in experimental diabetes induced by streptozotocin (author's transl)]. Yakugaku zasshi: Journal of the Pharmaceutical Society of Japan, (1981); 101(1): 86-90.
- Mustafa G, Anis E, Ahmed S, Anis I, Ahmed H, et al. Lupene-Type Triterpenes from Periploca a phylla. Journal of natural products, (2000); 63(6): 881-883.
- Prasad S, Kalra N, Shukla Y. Hepatoprotective effects of lupeol and mango pulp extract of carcinogen induced alteration in Swiss albino mice. Molecular nutrition & food research, (2007); 51(3): 352-359.
- Harborne JB, Baxter H, Webster FX. Phytochemical Dictionary: A Handbook of Bioactive Compounds from Plants. Journal of Chemical Ecology, (1994); 20(3): 815-818.
- Suzuki YA, Murata Y, Inui H, Sugiura M, Nakano Y. Triterpene glycosides of Siraitia grosvenori inhibit rat intestinal maltase and suppress the rise in blood glucose level after a single oral administration of maltose in rats. Journal of agricultural and food chemistry, (2005); 53(8): 2941-2946.
- Joo C, Kim J. Study on the hypoglycemic action of ginseng saponin on streptozotocin induced diabetic rats (I). Korean J Ginseng Sci, (1992); 16190-197.
- Basnet P, Kadota S, Shimizu M, Namba T. Bellidifolin: a potent hypoglycemic agent in streptozotocin (STZ)-induced diabetic rats from Swertia japonica. Planta medica, (1994); 60(06): 507-511.
- KARDONO LBS, BASUKI T, TRIANADEWI R, KAWANISHI K. Chemical Constituents of Scleroderma aurantium Ⅰ: A New Triterpene, 3, 25-Dihydroxy-22-acetoxyl-lanosta-8, 23-diene. Eurasian Journal of Forest Research, (2002); 5(1): 33-37.
- Kimura H, Ogawa S, Katsube T, Jisaka M, Yokota K. Antiobese effects of novel saponins from edible seeds of Japanese horse chestnut (Aesculus turbinata BLUME) after treatment with wood ashes. Journal of agricultural and food chemistry, (2008); 56(12): 4783-4788.




