Research Article
Comparative study to assess coagulation abnormalities in breast cancer
Toraiz Ahmed1, Rana Shakil Ahmed2, Muhammad Usman Basharat1, Muhammad Hassan Mushtaq1, Shakira Sadiq Gill1, Abdur Rehman Khawaja3, Abdul Basit*4, Arman Khan4, Muhammad Farhan Khan4
Adv. life sci., vol. 1, no. 2, pp. 96-103, November 2013
*Corresponding author: Abdul Basit (E-mail: boss_basit333@hotmail.com)
Author Affiliations
2- College of Statistical and Actuarial Sciences, University of Punjab, Lahore – Pakistan
3- Allama Iqbal Medical College, Jinnah hospital, Lahore – Pakistan
4- Institute of Biochemistry and Biotechnology University of Veterinary and Animal Sciences, Lahore – Pakistan
Abstract
Introduction
Methods
Results
Discussion
References
Abstract
Background: Coagulation abnormalities such as thrombosis and disseminated intravascular coagulation (DIC) are the major factors that play a major role in breast cancer. In this study, coagulation abnormalities were assessed in breast cancer patients to help the clinician in early detection of DIC and management of patients at different stages of breast cancer.
Methods: 75 patients were enrolled in the study, 50 were from case group (breast cancer patients) and 25 were selected as control group subjects used to compare the results. All of these subjects undergone, General Hematological analysis i.e. differential leukocyte count hemoglobin, platelets count and total leukocyte count were performed on each of the samples collected from the subjects and Specific Hematological analysis i.e. Activated Partial Thromboplastin Time (APTT), Fibrinogen Assay, Prothrombin Time (PT), D-Dimer Detection and Fibrin Degradation Products (FDPs).
Results: PT was found to be comparable in patients with breast cancer when compared with controls. Difference between control group (II) and subjects with breast cancer (I) was non-significant, fibrinogen level was found to be significantly increased (p < 0.01) in patients with different stages of breast cancer when compared with controls. FDPs were found to be significantly increased (p< 0.01) in patients of breast cancer when compared with control group. These increased levels of FDPs may be due to enhanced fibrinolysis. D-Dimers were also found to be significantly increased (p < 0.01) in patients with breast cancer when compared with controls.
Conclusion: Patients with breast cancer were associated with compensated DIC state including normal PT and APTT level but increased fibrinogen and platelets count as compared to the controls. Detection of D-Dimers offers a differential analysis over other laboratory tests for DIC.
Keywords: Disseminated intravascular coagulation, Prothrombin Time, Activated partial thromboplastin time, Fibrin Degradation Products, D-Dimers, Coagulation abnormalities, Breast cancer
Introduction
The coagulation disorders of non-haematological malignancies can cause either excessive clotting and bleeding or the combined abnormalities of excessive clotting direct to utilization of coagulation factors and secondarily guide to excessive bleeding. The non-haematological malignancies include carcinomas e.g. breast, prostate, ovary, lung, stomach tumors [1,2].
The essential role of the coagulation mechanism is carried out by the coagulation factors, coagulation inhibitory factors and fibrinolytic systems [3]. A bleeding tendency occurs when there is a deficiency of clotting factors or inhibition of coagulation mechanism. Coagulation factors play key role in haemostasis. Damage to the vessel wall results in simultaneous activation of platelets and the coagulation mechanisms. Platelet aggregation may be sufficient to occlude the damaged vessels temporarily but it needs fibrin, generated by coagulation mechanism for its permanent stability [4].
The derivatives of cross-linked fibrin are known as Plasma D-dimers. Production or formation of D-dimers depends upon the fibrin degradation by plasmin and the concentration increase as a result of thrombolysis. Plasma D-dimer represents a significant advance over current and historical FDP assays for diagnosis. The major indicators for the detection of plasma D-dimers are cross-linked fibrin derivatives devoid of intrusion from non-cross-linked fibrin/fibrinogen which consequently recognize fibrinolysis and intravascular thrombosis as different from fibrinogenolysis. Due to high credibility of D-dimer, monoclonal antibodies can be used with plasma samples, thereby distinguishing fibrinolysis from fibrinogenolysis and advising an advantage over most usual assays for FDP [5].
The only test that directly addresses both plasmin generation and thrombin is D-Dimer test [6]. The use of D-dimer test for diagnosis is effective and sensitive. During the management of DIC and thrombolytic therapy D-Dimer test may provide a good understanding of the creation and disbanding of fibrin in thrombotic disease [7]. Revealing of fragment DD, hence offers a exceptional lead over other laboratory tests for DIC, because it reports both dimensions of DIC [6].
D1C is normally characterized by consumption of platelets, blood clotting factors V and VIII, fibrinogen that may lead to decrease in their circulatory levels. This only occurs if the process is sufficiently acute. FDP circulatory levels also increase when they are produced as a result of secondary or compensatory fibrinolysis [8,9]. In about 5-10% of patients with micro thrombotic lesions, fibrin thrombi are frequently detected in micro arterioles of the glomeruli and might precipitate acute renal failure, a variety of thrombotic skin lesions and acute respiratory distress syndrome, may also occur if these thrombi occlude pulmonary vasculature. High levels of circulating fibrin complexes and FDP’s inhibit the action of thrombin and also stop platelet function, leading to continuing widespread bleeding tendency [10]. The D-dimer and FDP tests are highly effective for the express diagnosis of DIC [11,12].
Thrombosis in cancer is frequently migratory and involves superficial veins at somewhat typical positions. In the laboratory, indication of intravascular coagulation is readily found. Some have thrombocytopenia, indicated by increased level of FDP and reduction in the levels of fibrinogen, but these markers only correspond to the failure form of DIC, and a great volume reveals compensated or over compensated DIC. Thrombocytosis is more common, accompanied by decrease adhesion, aggregation and clot renunciation may also be experiential [13].
Non-specific mechanisms i.e. damage to the tissue and inflammatory reaction may result in the expression of tissue factors (TF). The tumor may also induce the expression of TF due to monocytes and tissue macrophages or express procoagulant activity (PCA) directly, giving them the capacity to produce thrombin even in the presence of other coagulation proteins. It has been demonstrated that TF expression is elevated in breast cancer patients [14]. Aside from the traditional members of the coagulation pathway, other prothrombotic mediators have been identified in tumor cells and include cancer procoagulants (PC) which are cysteine protease that activate factor X [15]. TF mediated tumor metastasis may affect translation of angiogenic growth factors i.e. vascular endothelial growth factor (VEGF).TF expression may also be increased by hypoxic conditions that act as a stimulus for VEGF production [16].
The APTT and PT may be expanded or reduced; hyperfibrinogenemia is common but acquired dysfibrinogenemia is rare unless the liver is involved. Cancer patients may have shortened euglobulin clot, decreased plasminogen levels, increase in plasmin-antiplasmin complexes and lysis time because of the co-occurrence of intravascular activation of coagulation and fibrinolysis. Most cancer patients having complications by sudden DIC have increased levels of FDP. Probability of amplified FDP is higher in cases with inaccessible metastases, matched to cancer confined to a small area and may have predictive rates. Plasma fibrinopeptide A (FpA) is prominent in most cases with malignant disease, and serial measurements may reflect tumor succession or reaction to management process [13]. Fibrinogenolysis and fibrinolysis cause the dissolution of circulating or precipitated fibrinogen and fibrin. Plasminogen is converted to plasmin, which is then able by hydrolysis to cleave portions of factors V and VIII, fibrinogen and fibrin, when in excess, this process creates hypocoagulable state. Fibrinogen and fibrin give up their respective degradation products FDPs and D-dimers [17].
Cancer patients may also suffer from the impediment of hemolytic uremic syndrome or thrombotic thrombocytopenic purpura (HUS/TTP). Renal failure, micro-angiopathic hemolytic anemia and severe thrombocytopenia should be considered significant indications in the characterization of HUS/TTP. Pulmonary symptoms i.e. edema in lungs is a regular examination. Disorders regarding nervous system are also often observed. The hemolysis can be gigantic and is because of red cell disintegration as schistocytes are observed in every case [18]. Patients with malignancy associated with DIC, DVT and thrombocytopenia suffers from hemostatic abnormalities [19].
The increased coagulable condition or "hypercoagulable state" of malignancy is because of a multifaceted interface of abnormal cells and their products with host cells; escorting to diverse extent of mutilation of the common defense system that usually guards the host in opposition to thrombogenesis. Uncontrolled cells can stimulate procoagulant properties and restrain anticoagulant properties of platelets monocytes, macrophages and vascular endothelial cells or can trigger straight forwardly the blood clotting mechanism and result in thrombosis. The pathophysiological local systematic findings collectively made make cancer conceivably the paramount illustration of "acquired thrombophilia [3,8].
In this study, main aims were to assess the coagulation abnormalities in patients of breast cancer and to help the clinician in early detection of DIC and management of patients at different stages of breast cancer.
Methods
Patients
For the study subjects were selected from, Institute of Nuclear Medicine and Oncology (INMOL) and Mayo Hospital, Lahore. Total 75 subjects were included in the study. The selected subjects were divided into two groups. Group I (case group) Inclusion Criteria: It included 50 breast cancer patients, all patients with breast cancer and patients without the personal and family history of bleeding diathesis. Exclusion Criteria: Patients with all hematological malignancies (leukemia, lymphoma, multiple myeloma), family history of bleeding diathesis, liver disease, hypertension, diabetes mellitus, renal disease and patients having history of chemotherapy and radio therapy. Group II (control group): It included 25 healthy subjects with age, sex and socio-economically matched controls from hospital staff or relative of the patients. They did not have any history of liver disease, diabetes mellitus or bleeding diathesis.
Laboratory Investigations
The blood samples were analyzed for general haematological investigations and coagulation profile. General Hematological Investigations: Hemoglobin, total leukocyte count, differential leukocyte count, platelets count are performed on the samples collected from the subjects. Specific Hematological investigations: PT, APTT, Fibrinogen Assay, FDPs, D-Dimer Detection was also done for collected samples. Data was managed and analyzed by using statistical software packages for social sciences (SPSS). For categorical variables, different codes were assign to them. Arithmetic mean (x), Standard Deviation (SD), Students‘t’ test and Probability Value (P value) analysis was performed.
Results
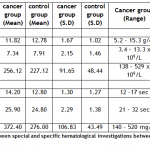
General Laboratory Investigations: In control group, the mean ± SD value of Hb was 12.78 ± 1.02 with a range of 11.5 – 14.3 g/dl and in subjects with breast cancer, the mean ± SD value of Hb was 11.82 ± 1.67 with a range of 5.2 – 15.3g/dl. On comparison between group II and group I, subjects with breast cancer showed decreased Hb level and the difference was greatly significant (p < 0.01) statistically. The mean ± SD values of TLC in controls was 7.91 ± 1.46 with a range of 4.5 – 10.2 x 109/L and in subjects with breast cancer was 7.34 ± 2.15 with a range of 3.4 – 13.3 x 109/L statistically. The comparison between group II and I was non-significant. The mean ± SD values of platelet count in control group (II) was 227.12 ± 48.44 with range of 154 – 329 x 109/L and in subjects with breast cancer (I), 256.12 ± 91.65 with a range of 138 – 529 x 109/L. When comparing these groups, Group I showed increased platelets count and difference was significant (P < 0.05) statistically. Special Laboratory Investigations: The mean ± SD values of PT in control group (II) was 12.8 ± 1.27 with range of 11 – 15 seconds and in group I (subjects with breast cancer) was 14.20 ± 1.30 with a range of 12 – 17 seconds. On comparison with control (II) PT was comparable in patients with breast cancer (Group I). The mean ± SD values of APTT in control group (II) was 24.80 ± 1.38 with range of 21 – 27 seconds and in Group I (subject with breast cancer) was 25.90 ± 2.90 with a range of 21 – 32 seconds. When comparing with control, difference was non-significant. The mean ± SD values of fibrinogen in control group (II) was 276.00 ± 43.49 with a range of 210 – 350 mg/dl and in patients with breast cancer (I) was 372.40 ± 106.83 with range of 140 – 520 mg/dl. Comparison with control group showed increased levels of fibrinogen in patients with breast cancer and difference was highly significant statistically (p < 0.01) (see table I).
In this study, control group (II) had 100% FDP levels in the range of < 5 µg/ml. In group I (patients with breast cancer), 15 out of 50 subjects (30%) had FDP levels of > 10 £ 40 µg/ml, 28 out of 50 subjects (56%) had levels in the range of ³ 40 µg/ml while only 7 out of 50 subjects (14%) had < 5 µg/ml. The FDP levels were found to be increased in subjects with breast cancer (Group I) as compared with control group (II) and the difference was statistically highly significant (p < 0.01). D-Dimers in all control group subjects were in the range of < 250 ng/ml. In patients with breast cancer (Group I), 29 out of 50 subjects (58%) had D-Dimer level in the range of 250 – 500 ng/ml, 12 out of 50 (24%) had levels in the range of 500 – 1000 µg/ml, 08 out of 50 (16%) had levels in the range of 1000 – 2000 ng/ml and only 01 out of 50 (02%) had levels in the range of < 250 ng/ml. D-Dimers level was found to be increased in subjects with breast cancer (Group I) as compared with control group (II) and difference was highly significant (p < 0.01) statistically. Histopathological Types in Patients with breast Cancer: 45 (90%) patients had invasive ductal carcinoma while 3 (6%) have invasive lobular carcinoma, 1 (2%) has mucinous and 1 (2%) papillary carcinoma. So this study shows that invasive ductal carcinoma is the most common type of breast cancer. FDPs in different stages of breast cancer: Out of 50 cases 7 (14%) cases had FDPs levels <5mg/ml i.e., 3 (6%) subjects with stage II and (8%), means 4 patients were in stage III. 15 (30%). Out of 50 patients having breast cancer, only 1 (2%) case had D-dimer level < 250ng/ml and 30 (60%) patients have D-dimers level 250-500ng/ml with 6 (18%) in stage II and 3 (6%) in stage IV. 11(22%) patients having D-dimers level 500-1000ng/ml with 6 (12%) in stage III and 5 (10%) in stage IV. 8(16%) patients having breast cancer had D-dimers level 1000-2000 ng/ml with 1 (2%) in stage III and 7(14%) in stage IV. When specificity of FDPs and D-dimers was compared, D-dimers were found to be more specific test in the detection of coagulation abnormalities in breast cancer patients because out of 7 (14%) patients with normal range of FDPs i.e., < 5mg/ml in stage II and III had raised levels of D-dimers (250-500ng/ml) in 6 (12%) cases.
Discussion
During the study Hb was found to be slightly lower in subjects with breast cancer as compared to control group (II) and difference was significant statistically. This decreased Hb may be due to the hemolytic process going on during the shedding of cells from the primary tumor and causing DIC. The findings are in agreement with that of [13] who also observed low Hb levels in subjects suffering from breast cancer. TLC in this study was found to be lower in subjects with breast cancer when compared with controls though it was non-significant statistically. Platelet count was found to be significantly higher (p < 0.05) in subject with breast cancer cases when compared with control group. This increased platelet count in breast cancer patients may be due to over compensated DIC. These results are in consistent with the findings of a pervious study [20] in which these changes were observed in breast cancer patients. PT was found to be comparable in patients with breast cancer when compared with controls. In this study, difference between control group (II) and subjects with breast cancer (I) was non-significant. Fibrinogen level was found to be significantly increased (p < 0.01) in patients with different stages of breast cancer when compared with controls. This increased fibrinogen level may be due to pro-coagulant effect of tumor tissue. This study is in agreement with the findings of [15] who also observed these changes in breast cancer patients. FDPs were found to be significantly increased (p< 0.01) in patients of breast cancer when compared with control group. These increased levels of FDPs may be due to enhanced fibrinolysis. These findings are consistent with the results of many investigators, including [8,15,20]. They observed increased levels of FDPs in women with breast cancer. In this study, D-Dimers were found to be significantly increased (p < 0.01) in patients with breast cancer when compared with controls. These increased levels of D-Dimers may be due to enhanced fibrinolysis. These findings are consistent with the results of [8,15]. They also observed similar increase of D-Dimers in different stages of breast cancer. Further case control and cohort studies are conducted on the same lines that may give helpful findings for the clinicians in early detection of breast cancer and DIC. Cohort studies in this regard better results for the research but they are to be costly as compared to case control studies. Early detection system for different disease could save number of years of life lost as in result of any disease related to genetic or cellular disorder [21, 22].
Data and Table/s
References
- Levi M, Ten Cate H. Disseminated intravascular coagulation. New England Journal of Medicine, (1999); 341(8): 586-592.
- Kumar R, Gupta V. Disseminated intravascular coagulation: current concepts. The Indian Journal of Pediatrics, (2008); 75(7): 733-738.
- Dahlbäck B. Blood coagulation. The Lancet, (2000); 355(9215): 1627-1632.
- Gawaz M. Role of platelets in coronary thrombosis and reperfusion of ischemic myocardium. Cardiovascular research, (2004); 61(3): 498-511.
- Francalanci I, Comeglio P, Liotta AA, Cellai A, Fedi S, et al. D-dimer plasma levels during normal pregnancy measured by specific ELISA. International Journal of Clinical and Laboratory Research, (1997); 27(1): 65-67.
- Lane D, Preston F, Ross M, Kakkar V. Characterization of serum fibrinogen and fibrin fragments produced during disseminated intravascular coagulation. British journal of haematology, (1978); 40(4): 609-615.
- Carr J, McKinney M, McDonagh J. Diagnosis of disseminated intravascular coagulation. Role of D-dimer. American journal of clinical pathology, (1989); 91(3): 280-287.
- Falanga A, Rickles FR. Pathophysiology of the thrombophilic state in the cancer patient; 1999. Copyright© 1999 by Thieme Medical Publishers, Inc. pp. 173-182.
- Caine GJ, Stonelake PS, Lip GY, Kehoe ST. The hypercoagulable state of malignancy: pathogenesis and current debate. Neoplasia (New York, NY), (2002); 4(6): 465.
- Flier JS, Underhill LH, Coller BS. Platelets and thrombolytic therapy. New England Journal of Medicine, (1990); 322(1): 33-42.
- Yu M, Nardella A, Pechet L. Screening tests of disseminated intravascular coagulation: guidelines for rapid and specific laboratory diagnosis. Critical care medicine, (2000); 28(6): 1777-1780.
- Adam SS, Key NS, Greenberg CS. D-dimer antigen: current concepts and future prospects. Blood, (2009); 113(13): 2878-2887.
- Rickles FR, Levine M, Edwards RL. Hemostatic alterations in cancer patients. Cancer and Metastasis Reviews, (1992); 11(3-4): 237-248.
- Edwards R, Silver J, Rickles F. Human tumor procoagulants: registry of the Subcommittee on Haemostasis and Malignancy of the Scientific and Standardization Committee, International Society on Thrombosis and Haemostasis. Thrombosis and haemostasis, (1993); 69(2): 205.
- Gordon SG, Franks JJ, Lewis B. Cancer procoagulant A: a factor X activating procoagulant from malignant tissue. Thrombosis research, (1975); 6(2): 127-137.
- Shoji M, Hancock WW, Abe K, Micko C, Casper KA, et al. Activation of coagulation and angiogenesis in cancer: immunohistochemical localization in situ of clotting proteins and vascular endothelial growth factor in human cancer. The American journal of pathology, (1998); 152(2): 399.
- Boersma R, Jie K-S, Verbon A, Van Pampus E, Schouten H. Thrombotic and infectious complications of central venous catheters in patients with hematological malignancies. Annals of oncology, (2008); 19(3): 433-442.
- Gordon LI, Kwaan HC. Thrombotic microangiopathy manifesting as thrombotic thrombocytopenic purpura/hemolytic uremic syndrome in the cancer patient; 1999. Copyright© 1999 by Thieme Medical Publishers, Inc. pp. 217-221.
- Carroll VA, Binder BR. The role of the plasminogen activation system in cancer; 1999. New York: Stratton Intercontinental Medical Book Corporation, c1974-. pp. 183-198.
- Francis J, Francis D, Gunathilagan G. Assessment of hypercoagulability in patients with cancer using the sonoclot analyzer™ and thromboelastography. Thrombosis research, (1994); 74(4): 335-346.
- Nazir S, Khan A, Nazar A, Fayyaz A, Khan MS, Ahmed S, Masood F, Yaqub T, Rakha A. Prevalence of syphilis in Pakistani blood donors. Advancements in life sciences, (2013): 1(1): 27-30.
- Ali N, Naeem S, Afridi SQ, Muhammad J, Bacha U. Molecular study of Apolipoprotein E gene in familial hypercholesterolemic families. Advancements in life sciences, (2013): 1(1): 13-17.