Full Length Research Article
Efficacy of Pelargonium graveolens essential oils against some postharvest fungal diseases of apple
Yassir Elouadi1, Hajar Bendaif2, Hamza Assaggaf3, Emad M Abdallah4*, Mouna Mekkaoui5, Hanae Naceiri Mrabti6, Mounir Manssouri1, Tauofiq Benali7, Abdelhakim Bouyahya8, Abdelhamid Bouyanzer1
Adv. life sci., vol. 9, no. 2, pp. 195-201, July 2022
*- Corresponding Author: Emad M Abdallah (Email: Emad100sdl@yahoo.com)
Authors' Affiliations
2. LCOMPN-URAC25, Faculty of Sciences, Laboratory of Organic Chemistry, Macromolecular and Natural Products, Mohamed 1st University, Oujda – Morocco
3. Department of Laboratory Medicine, Faculty of Applied Medical Sciences, Umm Al-Qura University, Makkah – Saudi Arabia
4. Department of Science Laboratories, College of Science and Arts, Qassim University, Ar Rass – Saudi Arabia
5. Pharmacodynamics Research Team ERP, Laboratory of Pharmacology and Toxicology, Faculty of Medicine and Pharmacy, University Mohammed V in Rabat – Morocco
6. Laboratory of Pharmacology and Toxicology, Bio Pharmaceutical and Toxicological Analysis Research Team, Faculty of Medicine and Pharmacy, University Mohammed V in Rabat – Morocco
7. Environment and Health Team, Polydisciplinary Faculty of Safi, Cadi Ayyad University, Sidi Bouzid – Morocco
8. Laboratory of Human Pathologies Biology, Department of Biology, Faculty of Sciences Mohammed V University in Rabat – Morocco
Abstract![]()
Introduction
Methods
Results
Discussion
References
Abstract
Background: Postharvest diseases that occur in apples are often caused by various pathogenic fungi, causing serious economic loss. The pathogenic fungi Penicillium expansum, Rhizopus stolonifer and Botrytis cinerea are among the most common pathogens in apples. The goal of this study was to see whether Moroccan Pelargonium graveolens essential oil (PGEO) could protect apple fruits from fungal infections after they were harvested (in vitro).
Methods: PGEO was characterized by GC-MS and for antifungal assessment, in vitro poisoned food (PF) and volatile activity testing (VA) were carried out.
Results: The investigation revealed that PGEO was effective against the three tested phytopathogenic fungi in a dose-dependent manner and this antifungal activity increased with the volatile activity test. The MIC value was 2 µL/mL for B. cinera, and R. stolonifer, and 1 µL/mL for P. expansum. Volatile fraction stops the growth of B. cinera at 40 µL / disc (QMI = 40 µL / disc), and of P. expansum and R. stolonifer at 80 µL / disc (QMI = 80 µL / disc).
Conclusion: The current findings show that Moroccan PGEO has powerful antifungal activities, suggesting that it might be used instead of synthetic fungicides to combat apple post-harvest infections.
Keywords: Pelargonium graveolens; Antifungal activity; Essential oil; Poisoned food; Volatile activity test; Apple
The apple fruit (Malus domestica Borkh.), is one of the most economically and culturally important nutritious fruits all over the globe; it grows in almost all temperate zones. The whole fruit is edible except the seeds; many other products are made from them, such as juice, jam, wine, cider tea, or dried apples [1]. Fungal diseases are thought to cause 5 to 25% losses in overall yield in apples during postharvest storage and marketing across the world [2]. The increasing incidences of plant-pathogenic fungal invasions of apples are thought to be an unintended consequence of globalization, climate change, and, more broadly, environmental damage [3]
The shelf life and conservation of fruit after harvest are largely influenced by parameters like temperature, humidity, microbial infection, and insect attacks. Generally, fruits are highly susceptible to fungal rot starting from the day of their flowering to the period of purchase by the consumer due to the short shelf life and maturity of the fruits. In the case of apples, the quantity and composition of the main components of natural surface waxes (hydrocarbons, alcohols, fatty acids, ursolic acid, and α-farnesene) change during storage [4]. The use of fungicides to control postharvest deterioration of fruits and vegetables has recently been severely limited. However, apples are sometimes treated with CaCl2 to lengthen their storage life and decrease postharvest deterioration [5]. These synthetic chemical fungicides cause severe consequences for the environment and human health. Accordingly, there is an intrinsic need to search for bio-compatibility fungicides with minimal side effects [6]. On the other side, multiple postharvest fungicide-resistant strains could be devastating for the fruit packing industry. Therefore, there is an intrinsic need to answer critical questions about the ability of these pathogenic fungi to develop resistance, the capacity to maintain resistance mechanisms, and the decay-causing possibilities of postharvest fungicide-treated apples [7].
Currently, several innovative techniques (including natural biocides, biological control agents, and stimulation of fruit defense systems) are emerging as potential synthetic fungicide alternatives [8]. Natural products are a rich source of bioactive ingredients with a wide range of applications as antimicrobial agents [9] in cosmetics [10] and in agricultural entomology and pest control [11]. Essential oils are volatile molecules extracted from aromatic herbs and are rich in bioactive phytochemical molecules. These volatile substances extracted from some herbs such as Salvia officinalis, Melissa officinalis, Calamintha nepeta and Thymus vulgaris have many protective advantages for plant tissues and fruits, like protection from pathogenic fungal attacks [12-15]. Blue mold caused by Penicillium expansum, gray mold caused by Botrytis cinerea, and rhizopic rot caused by Rhizopus stolonifer are three of the most common and destructive postharvest diseases in apples [16].
Pelargonium graveolens (P. graveolens), is an aromatic plant with a distinguished fragrant; it is known as Rose geranium, it belongs to the family Geraniaceae, distributed in many parts of Africa, Southern Asia, and Australia. Currently, grown in many countries in Europe, Asia and Madagascar for production of geranium oil [17]. The fragrant oils of P. graveolens are used in traditional medicine to treat inflammations, dysentery, hemorrhoids, menorrhagia, cancer, and are applied in skincare [18].
Moroccan traditional medicine is highly utilized, and multiple investigations into Moroccan medicinal plants have shown a variety of bioactive characteristics, including antibacterial [19] and antifungal activity [20]. The literature survey revealed that reports on essential oil of P. graveolens grown in Morocco are scant and accordingly. Therefore, the current study aimed (for the first time) to evaluate the possible antifungal efficacy of the Moroccan P. graveolens essential oils on some postharvest fungal illnesses of apple, as an alternative biocontrol substitute to synthetic fungicides.
Plant material and extraction
The leaves of P. graveolens grow wild in the Taza area, a northeastern region of Morocco. A taxonomist identified the plant, and a specimen voucher was deposited at the Oujda Faculty of Sciences herbarium, Morocco. The collected leaves have been dried in the shade for up to a week at 25-30C. Then, 100 grams of P. graveolens leaves were crushed to a fine powder and distilled in water for three hours with the aid of a Clevenger-type apparatus [21]. The resulting essential oil was dried with anhydrous sodium sulfate and kept in a dark bottle in the fridge.
Isolation of fungal strains
Three fungal species from rooted apples taken from different locations in the Midelt area at Morocco and three fungi have been isolated and identified with the routine technique for fungi identification [22] which were Rhizopus stolonifera Botrytis cinerea and Penicillium expansum. Fungal samples were transferred to sterile Petri dishes (9 cm) containing Potato Dextrose Agar (PDA) with streptomycin meant for stopping the growth of bacteria, and seeded plates were incubated for up to 7 days at 25 ± 2 °C in the dark. Then, a microscopic examination was performed following the protocol's description by Barnett and Hunter [23] and was subcultured again to ensure getting pure cultures, which were maintained at 4°C.
Poisoned food technique (PF)
The antifungal activity of the essential oil was evaluated using the poisoned food method [24]. The essential oil (0.2%) was emulsified in sterile hot PDA agar suspension and immediately added to a glass Petri dish (90 × 20 mm) before it gets cool, the tested concentrations of the essential oil (0.25 to 2 µL/mL) was mixed with the hot PDA for controls, a negative control was also prepared. Plates were injected with 6 mm mycelium plugs of and kept in the incubator for up to 7 days for P. expansum, 60 hours for R. stolonifer and 11 days for B. cinera at 25±2°C.
Volatile activity test (VA)
The antifungal activity of natural volatile organic molecules against pathogenic fungi was evaluated using VA as mentioned in Xing et al [25] with some modifications. Petri-dishes were packed with 20 mL of potato dextrose agar (PDA). After that, plates were inoculated with a mycelial disc (size: 6 mm), it was taken from the perimeter of the mycelium culture (grown for upto 7 days) of the tested mushrooms. The Petri dishes (20×90 mm) contained 80 mL of air space after adding 20 mL PDA medium. Plates were inverted upside down and sterilized filter paper discs (9 mm in diameter) and were impregnated with variable doses of essential oil at 10, 20, 40, 80, and 160 µL/disc, and then the inverted Petri-dishes were incubated for up to 48 hours for R. stolonifer, 6 days for P. expansum and 10 days for B. cinera at 25±2°C. Following the above-mentioned methods, for every test (fungus/quantity), three replicates were inoculated, and the mycelial growth was recorded by measuring the size of diameter along two vertical lines passing through the center of the Petri-dish. The fungi-toxicity of the tested essential oil was evaluated at a percentage of the inhibition of the mycelial growth of (1%) and calculated according to the formula [26]:
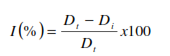
Where Dt and Di indicate the diameter of the mycelial growth in the control and the treated plates, respectively.
Minimum inhibitory concentration (MIC)
Minimal inhibitory concentrations (MIC) were also investigated using the procedures described by Mohammadi et al [27] after the volatile activity test (VA). When the mycelium of fungus reached the margins of the control dishes, the lowest concentration at which there was no trace of growth was identified as the minimal inhibitory concentration (MIC).
Antifungal EC50
EC50 is defined as the efficient dose that inhibited mycelium from growing by 50% The EC50 value was determined using the relation between the tested essential oil concentrations and the percentage of the fungal mycelial growth inhibition. The correlation between the fungicidal and the fungistatic behavior of the essential oil was evaluated by detecting a re-growth of the inhibited discs of mycelia after its transport to the untreated potato dextrose agar medium. When there is no growth, this is referred to as a fungicidal effect, and when there is the contrary, it is referred to as a fungistatic effect [28].
Data analysis
The analysis of variance tests (ANOVA) was used to statistically examine the data, and Tukey's HSD test was used to separate the means. The Statistical Analysis System software was employed in this study (SAS Institute Inc., USA).
Results
Composition of the essential oil
The chemical content of Pelargonium graveolens essential oils was previously examined in our labs and published [22], which recorded up to 40 organic compounds accounted for 80.4% of the overall essential oil. The main compounds were, citronellol (22.8%), isomenthone (13.2%), geraniol (6.4%), 10-epi-γ-eudesmol (6.1%), Z- rose oxide (4.7%), and citronellyl formate (4.6%).
Antifungal activity
According to the direct contact technique applied to the agar medium (poisoned food), the effects of the concentration of P. graveolens essential oils on the growth of the mycelium after an incubation time of B. cinerea (11 days), P. expansum (7 days) and R. stolonifer (60 hours) at 25 ± 2 °C are listed in Table 1.
These data revealed that the percentage of mycelial growth inhibition increases with the concentration of P. graveolens essential oils for each tested strain. This implies that this essential oil shows a significant activity of (p <0.05). It was clear that B. cinera and R. stolonifer showed great sensibility towards the PGEO at a 2 µL / mL concentration and 1 µL / mL for P. expansum. Examination of table 2 shows that the MIC value of P. graveolens against B. cinera and R. stolonifer is 2 µL/mL, and 1 µL/mL for P. expansum.
Furthermore, it is necessary to be mindful of the essential oil's fungitoxic nature (at 2 µL/mL for R. stolonifer and B. cinerea and, and 1 µL/mL for P. expansum). The displacement of a mycelium disc from potato dextrose agar medium with 2 µL/mL of fresh essential oil onto potato dextrose agar medium indicated that the mycelium of P. expansum and B. cinera did not develop after seven days and eleven days of incubation, respectively. Table 2 shows that our essential oil has a fungicidal impact on the strains tested at 2 µL/mL, in contrast to R. stolonifer and P. expansum, which show fungistatic activity at 1 µL/mL.
From Table 3, we have observed that the inhibition percentage increases with the amount of P. graveolens essential oil in each fungal strain tested, indicating that the essential oil of P. graveolens may blocks the development of all fungal strains. In addition to this, P. graveolens essential oil owns a significant antifungal property that have direct effect on the growth of the mycelium of each strain tested (p < 0.05). Moreover, all of the investigated strains were sensitive to P. graveolens essential oil.
This volatile fraction inhibits the growth of B. cinera at 40 µL / disc, and of P. expansum and R. stolonifer at 80 µL / disc. Therefore, it’s obvious that B. cinera is very sensitive to this essential oil, followed by R. stolonifer and then P. expansum. Furthermore, we can affirm that PGEO stops growing the three fungi tested during the micro-atmosphere technique better than the direct contact technique, particularly for the case of B. cinera. Mycelial plates were moved to a PDA medium in the absence of essential oil after inhibition of growth with P. graveolens (Figure 1).
As shown in Figure 1, the growth of the B. cinera fungus starts on day 4 at a concentration of 40 µL / disc, whereas the concentrations of 80 and 160 µL/disc do not allow any increase in mycelium for up to 10 days. Therefore, PGEO displayed fungistatic activity at 40 µL/disc and fungicidal activity at 80 µL/disc. In the case of P. expansum, from the fourth day, we noticed fungal growth at 80 µL/disc, and none was detected after treated by the PGEO at 160 µL/disc. This essential oil therefore demonstrated fungicidal activity against P. expansum at 160 µL / disc and fungistatic activity at 80 µL / disc. Moreover, R. stolonifer growth of the mycelium was seen in the first twelve hours at the concentration of 80 µL / disc, whereas none was observed when adding EO at 160 µL / disc. This EO displays fungicidal activity towards R. stolonifer at 160 µL / disc and fungistatic activity at 80 µL / disc.
Figures & Tables
Post-harvest fungal infections cause severe loss in fruits, particularly apples, and lead to economic loss. Unfortunately, these diseases are still difficult to control, besides the excessive use of pesticides and fungicidal materials has harmful impacts on the consumer and the environment despite their high inhibitory efficacy. Therefore, natural fungicides in particularly those extracted from medicinal plants, should be explored. In this context, volatile compounds (essential oils) have been the subject of several studies [29-31] Indeed, essential oils (EOs) can offer possible alternatives to the control of currently employed agents because that they are very rich in bioactive phytochemical compounds. Moreover, the use of EOs compounds to combat post-harvest fruit diseases has become an urgent need.
Generally, the antifungal activity of an EO is strongly linked to its type, concentration and chemical content. However, it’s expected that this activity also depends on which compounds act synergistically or antagonistically. Hence, all the results obtained in the current research reveals that the essential oils of P. graveolens exhibit significant antifungal activity with respect to the tested fungi based on in vitro experiments using poisoned food and volatile activity tests. The current study reported that Moroccan PGEO is rich in citronellol (22.8%), isomenthone (13.2%), and geraniol (6.4%), which are the main constituents, and our findings were in harmony with previous studies [32,33]. Moreover, these main compounds were reported as significant antimicrobial agents against yeasts [34,35], which gives us the possibility of confirming that these constituents are liable for the high antifungal activity of PGEO against the studied fungi with a slight modulation of the other minority components via a synergistic mechanism.
The present study revealed that the enhancement of the antifungal action of the essential oils was directly linked to the rise in oil concentration when used against the three fungi tested (Tables 1 and 3). We also noticed that the antifungal potential against the pathogens tested was more potent with the volatile fraction of PGEO than with the direct contact technique on the PDA agar medium. This finding is in very good accordance with the previously published data [34-36]. The mode of action can be explained by the fact that some compounds can make the membranes more permeable by removing the outer membrane of the fungi [37,38], while others cause the fungi to grow less and change the shape of their cells [39]. The hydrophobic nature of EOs can be assigned as the explanation for these observations. This nature causes a weak solubility in the agar medium and water. As a result, PGEO in vapor form is more absorbable by fungal mycelium than direct contact on agar. The latter is due to the lipophilic nature of the tissues of the fungus and the high-water content of the PDA agar [40]. On another level, fairly high doses of EOs are needed to stop mycelial growth in the direct contact phase. In comparison, a remarkable concentration of the EO has undesirable effects on the taste and natural flavor of the apple (organoleptic properties) [41]. Thus, using our oil tested in the vapor phase appears to be an auspicious concept that could be used to fumigation to control various post-harvest infections in the food industry, according to many published reports, several essential oils of medicinal plants may be used as a sustainable and efficient alternative to create new natural fungicides [42-44] .
Based on numerous published reports, Scientists have found that using natural antifungal compounds to protect fruits from phytopathogens after harvest is a promising trend. It was cited that, the use of natural essential oils including menthol, eugenol or thymol in a mixture with active packaging is an alternative to the use of chemical fungicides on fruits and vegetables, particularly those with a limited shelf life [45], Propolis is a naturally occurring resin collected from the leaves and bark of conifer and poplar trees. It has been shown that propolis can inhibit the fungal diseases caused by B. cinerea and P. expansum from spreading after a harvest of fruit and vegetables [46]. Recent interest in nanotechnology has grown, and it has been suggested as an alternative and effective system for managing fungal infections, since nanoparticles can readily enter fungal cells, inhibiting their growth and preventing illness. When nanomaterials are mixed with other chemicals, such as chitosan, their activity is enhanced [47]. Finally, the current results show that PGEO is effective against Rhizopus stolonifer, Penicillium expansum and Botrytis cinerea.
In conclusion, the current results support that PGEO are effective antifungal agents against Botrytis cinerea, Penicillium expansum, and Rhizopus stolonifer. The high antifungal activity of PGEO can be explained by a mix of multiple ways of acting and the intervention of different cellular targets. Taking into account the variety of molecules contained in the EO. Moreover, essential oils are characterized by their low toxicity, biodegradability, and they do not persist in the environment. According to these advantages, essential oil is regarded as potential alternatives to synthetic fungicides to protect apples from phytopathogenic fungi and prevent spoilage of other food products during storage. Evaluation of the antifungal activity of P. graveolens against phytopathogenic fungi in vivo is mandatory before their marketing.
Funding Statement
The authors would like to thank the Deanship of Scientific Research at Umm Al-Qura University for supporting this work by Grant Code: (22UQU4331100DSR01).
Author Contributions
The author declares that there is no conflict of interest regarding the publication of this paper.
References
- Patocka J, Bhardwaj K, Klimova B, Nepovimova E, Wu Q, et al. Malus domestica: A review on nutritional features, chemical composition, traditional and medicinal value. Plants, (2020); 9(11): 1408.
- Konstantinou S, Karaoglanidis G, Bardas G, Minas I, Doukas E, et al. Postharvest fruit rots of apple in Greece: Pathogen incidence and relationships between fruit quality parameters, cultivar susceptibility, and patulin production. Plant Disease, (2011); 95(6): 666-672.
- Gladieux P, Zhang X-G, Afoufa-Bastien D, Valdebenito Sanhueza R-M, Sbaghi M, et al. On the origin and spread of the scab disease of apple: out of central Asia. PloS one, (2008); 3(1): e1455.
- Manssouri M, Znini M, El Harrak A, Majidi L. Antifungal activity of essential oil from the fruits of Ammodaucus leucotrichus Coss. & Dur., in liquid and vapour phase against postharvest phytopathogenic fungi in apples. Journal of Applied Pharmaceutical Science, (2016); 6(5): 131-136.
- Moline HE, Locke JC. Comparing neem seed oil with calcium chloride and fungicides for controlling postharvest apple decay. HortScience, (1993); 28(7): 719-720.
- Sapper M, Palou L, Pérez-Gago MB, Chiralt A. Antifungal starch–gellan edible coatings with thyme essential oil for the postharvest preservation of apple and persimmon. Coatings, (2019); 9(5): 333.
- Jurick WM, Macarisin O, Gaskins VL, Park E, Yu J, et al. Characterization of postharvest fungicide-resistant Botrytis cinerea isolates from commercially stored apple fruit. Phytopathology, (2017); 107(3): 362-368.
- Jijakli M, Lepoivre P (2004) State of the art and challenges of post-harvest disease management in apples. Fruit and vegetable diseases: Springer. pp. 59-94.
- Abdallah EM. Plants: An alternative source for antimicrobials. Journal of Applied Pharmaceutical Science, (2011); 1(6): 16-20.
- Fonseca-Santos B, Corrêa MA, Chorilli M. Sustainability, natural and organic cosmetics: consumer, products, efficacy, toxicological and regulatory considerations. Brazilian Journal of Pharmaceutical Sciences, (2015); 5117-26.
- Cantrell CL, Dayan FE, Duke SO. Natural products as sources for new pesticides. Journal of natural products, (2012); 75(6): 1231-1242.
- El Ouadi Y, Manssouri M, Bouyanzer A, Majidi L, Lahhit N, et al. Essential oil composition and antifungal activity of Salvia officinalis originating from North-East Morocco, against postharvest phytopathogenic fungi in apples. Der Pharma Chemica, (2015); 7(9): 95-102.
- El Ouadi Y, Manssouri M, Bouyanzer A, Majidi L, Bendaif H, et al. Essential oil composition and antifungal activity of Melissa officinalis originating from north-Est Morocco, against postharvest phytopathogenic fungi in apples. Microbial pathogenesis, (2017); 107321-326.
- Ambrico A, Trupo M, Martino M, Sharma N. Essential oil of Calamintha nepeta (L.) Savi subsp. nepeta is a potential control agent for some postharvest fruit diseases. Organic Agriculture, (2020); 10(1): 35-48.
- Alamri SA, Hashem M, Alqahtani MS, Alshehri AM, Mohamed ZA, et al. Formulation of mint and thyme essential oils with Arabic gum and Tween to enhance their efficiency in the control of postharvest rots of peach fruit. Canadian Journal of Plant Pathology, (2020); 42(3): 330-343.
- Ewekeye T, Oke O, Esan O. Studies on post harvest rot of apple (Malus domestica Borkh). Indian Journal of Plant Sciences, (2016); 5(1): 36-41.
- Rana VS, Juyal JP, Blazquez MA. Chemical constituents of essential oil of Pelargonium graveolens leaves. International Journal of Aromatherapy, (2002); 12(4): 216-218.
- Sheppard J. Methods for routine detection of seedborne fungal pathogens. Journal of Seed Technology, (1979); 74-77.
- Doudach L, Al-Mijalli SH, Abdallah EM, Mrabti HN, Chibani F, et al. Antibacterial Evaluation of The Roots of Moroccan Aristolochia longa Against Referenced Gram-positive and Gram-negative Bacteria. Advancements in Life Sciences, (2022); 9(1): 116-121.
- Mekkaoui M, Assaggaf H, Qasem A, El-Shemi A, Abdallah EM, et al. Ethnopharmacological Survey and Comparative Study of the Healing Activity of Moroccan Thyme Honey and Its Mixture with Selected Essential Oils on Two Types of Wounds on Albino Rabbits. Foods, (2021); 11(1): 28.
- Ćavar S, Maksimović M. Antioxidant activity of essential oil and aqueous extract of Pelargonium graveolens L’Her. Food control, (2012); 23(1): 263-267.
- El Ouadi Y, Bouyanzer A, Majidi L, Paolini J, Desjobert J-M, et al. Evaluation of Pelargonium extract and oil as eco-friendly corrosion inhibitor for steel in acidic chloride solutions and pharmacological properties. Research on Chemical Intermediates, (2015); 41(10): 7125-7149.
- Barnett HL, Hunter BB. Illustrated genera of imperfect fungi. Illustrated genera of imperfect fungi, (1972); (3rd ed).
- Gakuubi MM, Maina AW, Wagacha JM. Antifungal activity of essential oil of Eucalyptus camaldulensis dehnh. against selected Fusarium spp. International journal of microbiology, (2017); 2017.
- Xing M, Zheng L, Deng Y, Xu D, Xi P, et al. Antifungal activity of natural volatile organic compounds against litchi downy blight pathogen Peronophythora litchii. Molecules, (2018); 23(2): 358.
- Pandey D, Tripathi N, Tripathi R, Dixit S. Fungitoxic and phytotoxic properties of the essential oil of Hyptis suaveolens/Fungitoxische und phytotoxische Eigenschaften des ätherischen Öis von Hyptis suaveolens. Zeitschrift für Pflanzenkrankheiten und Pflanzenschutz/Journal of Plant Diseases and Protection, (1982); 344-349.
- Mohammadi A, Hashemi M, Hosseini S. The control of Botrytis fruit rot in strawberry using combined treatments of Chitosan with Zataria multiflora or Cinnamomum zeylanicum essential oil. Journal of Food Science and Technology, (2015); 52(11): 7441-7448.
- Farzaneh M, Ahmadzadeh M, Hadian J, Tehrani AS. Chemical composition and antifungal activity of the essential oils of three species of Artemisia on some soil-borne phytopathogens. Communications in agricultural and applied biological sciences, (2006); 71(3 Pt B): 1327-1333.
- Montenegro I, Said B, Godoy P, Besoain X, Parra C, et al. Antifungal activity of essential oil and main components from Mentha pulegium growing wild on the Chilean central coast. Agronomy, (2020); 10(2): 254.
- Ma D, Ji D, Liu J, Xu Y, Chen T, et al. Efficacy of methyl thujate in inhibiting Penicillium expansum growth and possible mechanism involved. Postharvest Biology and Technology, (2020); 161111070.
- Mziouid A, Chebli B, Berrabah M, Heimeur N, Mayad E. Antifungal activities of essential oil from Moroccan endemic Anthemis tenuisecta and seed emergence. Materials Today: Proceedings, (2020); 27: 3108-3113.
- Boukhatem MN, Kameli A, Saidi F. Essential oil of Algerian rose-scented geranium (Pelargonium graveolens): Chemical composition and antimicrobial activity against food spoilage pathogens. Food control, (2013); 34(1): 208-213.
- Araujo C, Sousa M, Ferreira M, Leao C. Activity of essential oils from Mediterranean Lamiaceae species against food spoilage yeasts. Journal of food protection, (2003); 66(4): 625-632.
- Sharma N, Tripathi A. Fungitoxicity of the essential oil of Citrus sinensis on post-harvest pathogens. World Journal of Microbiology and Biotechnology, (2006); 22(6): 587-593.
- Survilienė E, Valiuškaitė A, Snieškienė V, Stankevičienė A. Effect of essential oils on fungi isolated from apples and vegetables. Sodininkystė ir daržininkystė, (2009); 28(3): 227-234.
- Soylu EM, Kurt Ş, Soylu S. In vitro and in vivo antifungal activities of the essential oils of various plants against tomato grey mould disease agent Botrytis cinerea. International Journal of Food Microbiology, (2010); 143(3): 183-189.
- Edris AE, Farrag ES. Antifungal activity of peppermint and sweet basil essential oils and their major aroma constituents on some plant pathogenic fungi from the vapor phase. Food/Nahrung, (2003); 47(2): 117-121.
- Burt S. Essential oils: their antibacterial properties and potential applications in foods—a review. International journal of food microbiology, (2004); 94(3): 223-253.
- Ziedan E, Farrag ES. Fumigation of peach fruits with essential oils to control postharvest decay. Research Journal of Agriculture and Biological Sciences, (2008); 4(5): 512-519.
- Helander IM, Alakomi H-L, Latva-Kala K, Mattila-Sandholm T, Pol I, et al. Characterization of the action of selected essential oil components on Gram-negative bacteria. Journal of agricultural and food chemistry, (1998); 46(9): 3590-3595.
- Lambert R, Skandamis PN, Coote PJ, Nychas GJ. A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. Journal of applied microbiology, (2001); 91(3): 453-462.
- Tripathi A, Sharma N, Sharma V, Alam A. A review on conventional and non-conventional methods to manage post-harvest diseases of perishables. Researcher, (2013); 5(6): 6-19.
- Kumar PS, Nattudurai G, Islam VIH, Ignacimuthu S. The effects of some essential oils on Alternaria alternata, a post-harvest phyto-pathogenic fungus in wheat by disrupting ergosterol biosynthesis. Phytoparasitica, (2022); 50(2): 513-525.
- Mounir M, Omar O-A, Lahcen O, Amal L, Abdeslam A, et al. Antifungal activity of essential oil from Santolina pectinata lag., against postharvest phytopathogenic fungi in apples. Arabian Journal of Medicinal and Aromatic Plants, (2022); 8(1): 41-54.
- Serrano M, Martinez-Romero D, Castillo S, Guillén F, Valero D. The use of natural antifungal compounds improves the beneficial effect of MAP in sweet cherry storage. Innovative food science & emerging technologies, (2005); 6(1): 115-123.
- Tripathi P, Dubey N. Exploitation of natural products as an alternative strategy to control postharvest fungal rotting of fruit and vegetables. Postharvest biology and Technology, (2004); 32(3): 235-245.
- González-Estrada R, Blancas-Benitez F, Moreno-Hernández C, Coronado-Partida L, Ledezma-Delgadillo A, et al. (2019) Nanotechnology: A promising alternative for the control of postharvest pathogens in fruits. Nanotechnology for Agriculture: Crop Production & Protection: Springer. pp. 323-337.
This work is licensed under a Creative Commons Attribution-Non Commercial 4.0 International License. To read the copy of this license please visit: https://creativecommons.org/licenses/by-nc/4.0








