Full Length Research Article
Response Surface Methodology for the production of endopolygalacturonase by a novel Bacillus licheniformis
Saqib Hussain Hadri1*, Muhammad Javaid Asad1*, Muhammad Zeeshan Hyder2, Syed Muhammad Saqlan Naqvi1, Tariq Mukhtar1, Raja Tahir Mehmood3, JH David Wu4
Adv. life sci., vol. 5, no. 4, pp. 204-210, August 2018
*- Corresponding Author: Saqib Hussain Hadri (Email: Saqib Hussain Hadri)
Authors' Affiliations
2- Department of Bioscience, COMSATS Institute of Information and Technology, Islamabad, Pakistan
3- Department of Biotechnology, Mirpur University of Science and Technology (MUST), Mirpur Azad Jamu and Kashmir, Pakistan
4- Department of Chemical Engineering, University of Rochester, Rochester, NY, USA
Abstract![]()
Introduction
Methods
Results
Discussion
References
Abstract
Background: Polygalacturonase is one of the most important commercial pectinase. The production cost and the mesophilic nature of the present polygalacturonase is a big problem in its application in the juice industry. A lot of work is going on for the isolation of thermophilic bacterial strains which can utilize pectin as the only carbon source.
Methods: Bacterial strains were isolated from rotten fruits and vegetables and cultured at 50 – 70oC. The strains were than screened for endopolygalacturonase activity and identified on the basis of 16S rRNA sequence. Different growth parameters for the production of endopolygalacturonase by Bacillus licheniformis IEB-8 were optimized using Response Surface Methodology under Center Composite Design using JMP-12 software. Endopolygalacturonase was purified in two steps; ammonium sulfate precipitation and then by size exclusion column chromatography.
Results: Only four strains, IEB-8, IEB-11, IEB-12 and IEB-13 showed growth above 60oC. Among these four, only IEB-8 was found to be endopolygalacturonase positive, which was identified as Bacillus licheniformis by 16S rRNA gene sequence. Purification fold of 2.57 and 7.48 in the specific activity were achieved using ammonium sulfate precipitation and gel filtration chromatography respectively. Molecular weight of the purified endopolygalacturonase was found to be 42 kDa. The purified endopolygalacturonase showed an optimum pH of 7 and optimum temperature of 55oC.
Conclusion: Bacillus licheniformis IEB-8 is a novel bacteria which can efficiently be utilized in the industry for the production of endopolygalacturonase very cheaply. Furthermore, the high optimum working temperature of endopolygalacturonase, increases its significance for its industrial applications.
Keywords: Endopolygalacturonase; Bacillus licheniformis; Thermophilic; Response Surface Methodology; Ammonium sulfate precipitation
Enzymes cleaving the pectic substances are broadly known as pectinolytic enzymes or pectinases [1]. These enzymes include endopolygalacturonase(endo-PG) (EC 3.2.1.15), exopolygalacturonase (exo-PG) (EC 3.2.1.67), pectatelyases (EC 4.2.2.2), pectin lyases (EC 4.2.2.10) and pectin methyl esterases (EC 3.1.1.11) [2-4]. Polygalacturonases (PGs) obtained from different sources work at different pH values, which are typically acidic or rarely basic, and determine the potential application of these enzymes [5]. The importance of these enzymes are reflected in their annual sales which is found to be $75 million [6]. Aspergillus niger is usually used for the commercial production of PGs on industrial scale. However, very few researches have been done for the production of PGs from bacterial sources [3]. Bacteria have extra advantage that they are not influenced by various climatic conditions and other seasonal factors [7]. Bacillus bacteria are considered as the most suitable source of many commercially used enzymes. They are advantageous because they can be cultured in lab under extreme conditions like high temperature and wide pH range, resulting in the production of industrially important enzymes, which are very stable and active in a wide range of harsh environmental conditions [8].
Recently, response surface methodology (RSM) has been extensively used for the process and media optimization studies for enzyme production [9,10]. This method represents a set of statistical techniques for executing planning and evaluating the effect of independent variables on the desired variable response [10]. Polygalacturonases that work at high temperature are very important for their applications in industrial process which are carried-out at 50-60°C [11]. Processing of fruits and vegetables at high temperature causes reduction of microbial contamination as well as increase the pectin solubility and decrease the viscosity of liquid [12]. A lot of work is going on for the isolation of thermophilic bacterial strains which can utilize pectin as the only carbon source [2].
Keeping these points in mind, the present study was carried out for the isolation, identification and characterization of a Bacillus species which can grow above 50oC and produce thermophilic endo-PG.
Isolation of the microorganism
Rotten fruits and vegetables were collected from Rawalpindi and Islamabad during summer season [13,14]. The rotten parts of these samples were dissolved in the normal saline solution (0.9% NaCl in distilled water) and spread on medium containing 2% agar, 1% yeast extract, 1% peptone and 0.5% sodium chloride (YEP medium) and grown at 50-70oC [15]. The isolates were purified by sub-culturing on the new plates. Four isolates, IEB-8, IEB-11, IEB-12 and IEB-13, showed growth above 60oC. The purified isolates were preserved in the form of STAB cultures at 4oC [16,17]and 50% glycerol stock solutions at -80oC [18, 13].
Screening for endo-polygalacturonase activity
Endo-polygalacturonase positive isolates were screened in three steps. In the first step, pectinase positive isolates were screened. For this purpose, 2µL of the isolates were grown at 55oC for 24 hours in the screening medium (YEP plus pectin) [19]. The plates were flooded with the 3% CTAB solution [20,21] and incubated at 55oC for 10-15 minutes. [15,22,21]. Pectinase positive cultures were indicated with a clear zone around colonies. In the second step polygalacturonase positive cultures were screened by the polygalacturonase assay [23,24,19,17,13,25]. The product of the polygalacturonase assay was run on the TLC plate. Galacturonic acid was used as standard to check the nature of the polygalacturonase to be endo- or exo-[26].
Molecular identification of the isolate
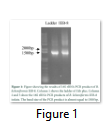
DNA was isolated from the liquid culture by CTAB method. [13,25]. Universal primers 27F (5’AGAGTTTGATCCTGGCTCAG3’) and 1492R (5’CGGTTACCTTGTTACGACTT3’) were used for the 16S rRNA gene amplification [3,25]. The PCR mixture contained 1µL of 10mM dNTPs, 5µL of 10X PCR buffer, 4µL of MgCl2 (50mM), 2µL of forward primer (10µM), 2µL of reverse primer (10µM) and Taq polymerase 0.25µL, 3µL of Genomic DNA and 32.75 µL of sterile double distilled water in a 50µL reaction mixture [22,19]. The PCR product was run on 0.5% agarose gel (containing 2µL of cyber stain) and confirmed by band almost equal to 1500bp (Figure 1). The PCR product was gel purified using “Ultra Clean® 15 DNA Purification Kit” by “MO BIO Laboratories, Inc.”. The purified PCR product was sequenced from “University of Rochester Center of Genomics, University of Rochester, NY, USA”. The sequence was assessed through “DNA Dragon 1.5.2” to obtain a consensus sequence and was submitted in the GenBank to get the accession number. The strain was identified by using the sequence of the 16S rRNA gene through EzBioCloud's Identify service (https://www.ezbiocloud.net/identify) and further searched using BLAST tool available at NCBI server (https://blast.ncbi.nlm.nih.gov/Blast.cgi) [19,22,13].
Response surface methodology for the optimization of the production parameters for endo-PG by Bacillus licheniformisIEB-8
Growth temperature, pH, inoculum size, inoculum age, substrate water ratio and fermentation period were optimized for IEB-8. Commonly, each parameter is being optimized separately [27,28,19,15,4], making the optimization a time consuming process. RSM under central composite design (CCD) was used for the optimization of all of the production parameters for the endo-PG [17]. RSM gave 46 treatments for the optimization of all of the said parameters in one experiment, and if doing separately, these parameters would have been optimized in seven days at minimum. JMP-12 was used for designing the experiment.
Use of agriculture waste for the production of the endo-PG
Citrus peels, white-radish peels and apple peels, being amongst the major agriculture wastes of the local area were used for the production of endo-PG. These substrates were collected in bulk quantities from the local market, dried and ground to fine powder. The powder was meshed with 40mm sieve [17,4]. This meshed powder was used as substrate for the growth of IEB-8, resulting in the production of different enzymes included endo-PG. The agriculture waste which gave the maximum production of endo-PG, was used in the next experiments.
Purification of endo-PG
Partial purification by ammonium sulfate precipitation
Different fractions of ammonium sulfate including 40%, 50%, 60%, 70% and 80% were used for the partial purification of the endo-PG. The calculated mass of ammonium sulfate was added slowly to the crude extract placed at 4oC [27,22]. The crude extract was continuously stirred to dissolve the added ammonium sulfate. The contents were allowed to stir for 15 minutes and centrifuged at 13000 RPM (20217 g) for 15 minutes. The supernatant was used for precipitation with the next percentage of the ammonium sulfate and the pellet was dissolved in the phosphate buffer (pKa7.2) for the membrane dialysis [29]. “Slide-A-Lyzer® 3.5K Dialysis Cassettes G2” by “Thermo Scientific” were used to dialyze the samples. All of the extracts were used for the enzyme assay to check the presence of the endo-PG [30,4].
Size exclusion column chromatography for the further purification
Partially purified endo-PG was further purified by size exclusion column chromatography. Sephadex G-100 was used for size exclusion column chromatography. The gel was packed in the 60×100 column by several runs of the buffer. One (1) mL of the partially purified and dialyzed extract was applied to the column for the purification. Fifty elusions were taken after the interval of 2 minutes [4]. All of the elusions were run on nano-drop spectrophotometer to check the presence of the proteins. The elusions giving the presence of the proteins were used for the endo-PG assay to check the presence of endo-PG. The elusion giving the positive endo-PG assay was run on SDS-polyacrylamide gel electrophoresis to check the purity of the sample [16,27,22,19].
Characterization of endopolygalacturonase
Endo-PG was characterized by determining its optimum temperature and pH of its activity. Enzyme assay was carried out at different temperatures (45oC, 50oC, 55oC, 60oC and 65oC) and different pH values (5, 6, 7, 8 and 9) of the buffer.
Molecular characterization of IEB-8 as B. licheniformis
The 16S rRNA gene sequence of B. licheniformis IEB-8 was submitted to GenBank with an accession number KY816931. The 16S rRNA gene sequence of IEB-8 was identified by EzBioCloud's Identify service (https://www.ezbiocloud.net/identify) using authentic quality-controlled databases of 16S rRNA sequences. The full-length 16S rRNA gene showed highest sequence identity of 99.86% with DSM 13 strain (ATCC 14580T) of Bacillus licheniformis (GenBank accession number AE017333) suggesting IEB-8 to be a strain of Bacillus licheniformis species. The BLAST search on NCBI nucleotide database [19,13], showed similarity with many of the B. licheniformis strains.
Optimization of production parameters using RSM
Fermentation period, inoculum size, inoculum age, substrate water ratio, growth temperature and growth pH were optimized using RSM under CCD using JMP-12 software. Enzyme activity was determined against each treatment and was inserted in the software. Surface profiler and contour profiler of the treatments were prepared for all of the parameters. Surface profiler (3D) and contour profiler (2D) [17] of the said parameters are shown in the Figure 2 and figure 3 respectively. Contour profiler showed the inoculum age to be 16.32 hours, inoculum size to be 7.77%, substrate water ratio to be 2.98%, fermentation period to be 70.58 hours, temperature to be 58.65oC and pH to be 6.97. All of the optimized parameters were also obtained from the prediction profiler shown in figure 4.
Purification of endo-PG
Ammonium sulfate partial precipitation of endo-PG
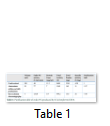
Among different concentrations of ammonium sulfate used for the partial purification of endo-PG, 70% gave the maximum precipitation. The maximum amount of endo-PG precipitated was checked using the endo-PG assay. The initial concentrations of ammonium sulfate did not show any of the considerable endo-PG activity, but 70% ammonium sulfate showed maximum endo-PG activity. The partially purified endo-PG showed endo-PG activity of 77 IU/mL/min and specific activity of 11 U/mg with a 2.57 fold increase in the specific activity (table 1).
Size exclusion column chromatography for the purification of endo-PG
Fifty elusions were taken from the column chromatography and were initially run on the nano-drop spectrophotometer at 280nm to check the presence of the proteins. Twenty five elusions showed the presence of the proteins and were used for the endo-PG assay. Out of those 25 elusions, only two elusions gave the positive assay for endo-PG. One of the elusions was run on the SDS-PAGE. The stained SDS-PAGE, showed one band indicating the presence of a single protein, i.e. endo-PG. The size of the endo-PG was found to be 42kDa (Figure 5). The gel purified endo-PG showed endo-PG activity of 124.8 U/mL/min and specific activity of 32 U/mg with a 7.48 fold increase in the specific activity (table 1).
Characterization of endo-PG
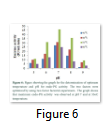
Among different pH values used for the enzyme assay, endo-PG gave good results at pH 6 and 7, while 55oC was proved to be the optimum temperature for the endo-PG activity. Increasing or decreasing temperature had negative effect on the endo-PG activity, similarly an increased or decreased pH than 7 also showed negative effects on the endo-PG activity (Figure 6). Optimum temperature of 55oC and pH of 7 showed an endo-PG activity of 46 IU/mL/min. Endo-PG showed activity of 36 IU/mL/min at pH 8 and 55oC indicating that it was somewhat stable at pH 8. Different optimum temperatures and pH values had been reported by different scientists.
Tables & Figures
Polygalacturonases that work at high temperature are very important for the processing of the fruit juices, which is carried-out at 50-60°C. Fungal PGs have optimum pH ranging from 3.5 to 5.5 [31] and being acidic in nature cannot be used for the maceration of vegetables while bacterial PGs being alkaline in nature can efficiently be utilized for the maceration of the vegetables. Most of the reported PG production is from mesophilic bacteria and ranges from 32-37oC [32]. Jahan et al. [33] reported the production of polygalacturonase from B. licheniformis KIBE-IB3 from agro-waste with a growth temperature of 37oC. Ghani et al. [34] reported the production of polygalacturonase from B. licheniformis at 37oC. Rehman et al. [14] and Embaby et al. [31] have reported the production of polygalacturonase from B. licheniformis KIBGE-IB21 and B. licheniformis SHG-10 respectively. All of them have reported the optimum growth temperature of the B. licheniformis to be 37oC. The current study resulted in the isolation of a novel thermophilic strain of B. licheniformis IEB-8 producing endopolygalacturonase, capable of growing over 60oC with an optimum growth at 58oC.
Commercialization of any enzyme is constricted to the high cost of production process. Efforts are going on to find the new ways to make the production cheap by using different agro-wastes. Rehman et al. [14] have mentioned the use of agro-waste as the sole source for the growth of the B. licheniformis SGH10. Jahan et al. [33] have mentioned the screening of different agro-wastes, including apple peels, orange peels, lemon peels, potato peels and wheat bran as source of nutrients for the production of pectinase by B. licheniformis KIBE-IB3, and finally mentioned wheat bran as best pectin source. In our study, we evaluated the potential of citrus peels, white-radish peels and apple peels as a source of pectin and other nutrients for the growth of B. licheiformisIEB-8. Citrus peels gave the maximum production of PG with an activity of 45 IU/mL/minute. B. licheniformis IEB-8 used citrus peels as the only source of nutrients.
Ammonium sulfate is preferred for the precipitation of proteins because of the low cost and rare denaturation of proteins. The optimum concentration of ammonium sulfate causing the precipitation of the proteins ranges from 60-80%. [35]. Ngo et al. [16] had mentioned 70% ammonium sulfate concentration to be the optimum for the endo-PG precipitation. Arijit et al. [22] have reported 80% of the ammonium sulfate concentration to be the best for pectinase precipitation with a 2.6 fold increase in the enzymatic activity. Different scientists have reported different molecular weight of PGs, which ranged from 38kDa to 72kDa [19,4]. The molecular weight of our extracted endo-PG falls in the already reported endo-PG’s molecular weight.
Different reports are available considering the optimum pH and temperature of the PG activity. PGs produced by the fungi have optimum pH in the acidic range while PG obtained from the bacteria have optimum pH in the neutral-alkaline scale [31]. Our results are in agreement with this statement with an optimum pH around 7 which is neutral-alkaline. Some other scientists also have reported optimum pH to be 6.5 [36,19].
Among different reports available for the production of PG by different bacteria, this is the only report, describing the production of endo-PG, having optimum activity at 55oC, which is the ideal temperature for its application in the industries. B. licheniformis IEB-8 is able to utilize citrus peels as a sole source of nutrients for its growth resulting in the cheap production of endo-PG. Ammonium sulfate and ammonium chloride further enhanced the production of endo-PG in the presence of citrus peels. The enzyme was characterized with an optimum pH around 7, optimum temperature to be 55 oC and specific activity of 32 U/mg.
Conflict of interest
Authors declare that there is no conflict of interest for publishing this study.
Acknowledgement
I present my gratitude to “Pir Mehr Ali Shah Arid Agriculture University, Rawalpindi” and “Higher Education Commission” Pakistan for the funding, which enabled me to carry out all of this research work in “University of Rochester, Rochester, NY, USA”. I am sincerely grateful to the lab members including Mr. Ryan, Mr. Yuan and admin staff of University of Rochester particularly Ms. Sandra for their help and guidance.
- Yang J, Luo H, Li J, Wang K, Cheng H, et al. Cloning, expression and characterization of an acidic endo-polygalacturonase from Bispora sp. MEY-1 and its potential application in juice clarification. Process Biochemistry, (2011); 46(1): 272-277.
- Saad N, Briand M, Gardarin C, Briand Y, Michaud P. Production, purification and characterization of an endopolygalacturonase from Mucor rouxii NRRL 1894. Enzyme and Microbial Technology, (2007); 41(6): 800-805.
- Khusro A, Aarti C. Discovery of polygalacturonase producing Bacillus tequilensis strain ARMATI using 16s rRNA gene sequencing. Asian Journal of Pharmaceutical and Clinical Research, (2015); 8(5): 58-62.
- Sethi B K, Nanda PK, Sahoo S. Enhanced production of pectinase by Aspergillus terreus NCFT 4269.10 using banana peels as substrate. 3 Biotech, (2016); 6(1): 1-15.
- Kubra KT, Ali S, Walait M, Sundus H. Potential applications of pectinases in food, agricultural and environmental sectors, (2018); 6(2): 23-34.
- Zaslona H, Trusek-Holownia A. Enhanced production of polygalacturonase in solid-state fermentation: Selection of the process conditions, isolation and partial characterization of the enzyme. Acta biochimica Polonica, (2014); 62(4): 651-657.
- Suneetha V, Prathyusha K. Bacterial pectinases and their potent biotechnological application in fruit processing/juice production industry: A review. Journal of Phytology, (2011); 3(6): 16-19
- Wang XJ, Bai JG, Liang YX. Optimization of multienzyme production by two mixed strains in solid-state fermentation. Applied Microbiology and Biotechnology, (2006); 73(3): 533-540.
- Amin F, Bhatti HN, Bhatti IA, Asgher M. Utilization of wheat bran for enhanced production of exopoligalacturonase by Penicillium notatum using response surface methodology. Pakistan Journal of Agricultural Sciences, (2013); 50(3): 469-477.
- Yadav S, Dubey AK, Anand G, Kumar R, Yadav D. Purification and biochemical characterization of an alkaline pectin lyase from Fusarium decemcellulare MTCC 2079 suitable for crotolaria juncea fiber retting. Journal of basic microbiology, (2014); 54(S1): 161-169
- El-Sheekh MM, Abdel-mohsen SI, El-Abd MA, Hegazy EM, El-Diwany AI. Effective technological pectinases by Aspergillus carneus NRC1 utilizing the egyptian orange juice industry scraps. International Biodeterioration & Biodegradation, (2009); 63(1): 12-18.
- Andrade MVVd, Delatorre AB, Ladeira SA, Martins MLL. Production and partial characterization of alkaline polygalacturonase secreted by thermophilic Bacillus sp. SMIA-2 under submerged culture using pectin and corn steep liquor. Food Science and Technology (Campinas), (2011); 31(1): 204-208.
- Khan F, Latif Z. Molecular characterization of polygalacturonase producing bacterial strains collected from different sources. JAPS, Journal of Animal and Plant Sciences, (2016); 26(3): 612-618.
- Rehman HU, Siddique NN, Aman A, Nawaz MA, Baloch AH et al. Morphological and molecular based identification of pectinase producing Bacillus licheniformis from rotten vegetable. Journal of Genetic Engineering and Biotechnology, (2015); 13(2): 139-144.
- Kaur S, Kaur HP, Prasad B, Bharti T. Production and optimization of pectinase by Bacillus sp. isolated from vegetable waste soil. Indo American Journal of Pharmaceutical Research, (2016); 6(1): 4185-4190.
- Ngo L, Pham T, Le V. Purification of endopolygalacturonase from submerged culture of Aspergillus awamori L1 using a two-step procedure: Enzyme precipitation and gel filtration. International Food Research Journal, (2008); 15(2): 135-140.
- Handa S, Sharma N, Pathania S. Multiple parameter optimization for maximization of pectinase production by Rhizopus sp. C4 under solid state fermentation. Fermentation, (2016); 2(2): 1-9.
- Garcia-Maceira FI, Di Pietro A, Huertas-Gonzalez MD, Ruiz-Roldan MC, Roncero MI. Molecular characterization of an endopolygalacturonase from Fusarium oxysporum expressed during early stages of infection. Applied and Environmental Microbiology, (2001); 67(5): 2191-2196.
- Paudel YP, Lin C, Shen Z, Qin W. Characterization of pectin depolymerising exo polygalacturonase by Bacillus sp. HD2 isolated from the gut of apis mellifera l. Microbiology Discovery, (2015); 3(1): 1-8.
- Namasivayam E, Ravindar J, Mariappan K, Akhil J, Mukesh K et al. Production of extracellular pectinase by Bacillus cereus isolated from market solid waste. Journal of Bioanalysis & Biomedicine, (2011); 3(3): 70-75.
- Mehta S, Mitali R, Nilofer S, Nimisha P. Optimization of physiological parameters for pectinase production from soil isolates and its applications in fruit juice clarification. Journal of Environmental Research and Development, (2013); 7(4A): 1539-1546
- Arijit D, Sourav B, Naimisha R, Rajan S. Improved production and purification of pectinase from Streptomyces sp. GHBA10 isolated from valapattanam mangrove habitat, kerala, india. International Research Journal of Biological Sciences, (2013); 2(3): 16-22.
- Torres S, Sayago JE, Ordoñez RM, Isla MI. A colorimetric method to quantify endo-polygalacturonase activity. Enzyme and microbial technology, (2011); 48(2): 123-128.
- Gomes E, Leite RSR, Da Silva R, Silva D. Purification of an exopolygalacturonase from Penicillium viridicatum RFC3 produced in submerged fermentation. International journal of microbiology, (2009); 631942.
- Sohail M, Latif Z. Phylogenetic analysis of polygalacturonase-producing Bacillus and Pseudomonas isolated from plant waste material. Jundishapur journal of microbiology, (2016); 9(1): e28594.
- Saito K, Takakuwa N, Oda Y. Purification of the extracellular pectinolytic enzyme from the fungus Rhizopus oryzae NBRC 4707. Microbiological Research, (2004); 159(1): 83-86.
- Kar S, Ray RC. Purification, characterization and application of thermostable exo‐polygalacturonase from Streptomyces erumpens MTCC 7317. Journal of food biochemistry, (2011); 35(1): 133-147.
- Dharmik P, Gomashe A. Bacterial polygalacturonase (pg) production from agro industrial waste by solid state fermentation. Indian Journal of Applied Research, (2013); 3(6): 439-442.
- Al-Rajhi AM. Purification and characterization of an extracellular poly-galacturonase from Rhizoctonia solani KÜHN (AG2-2). World Applied Sciences Journal, (2013); 21(4): 476-484.
- Udenwobele DI, Nsude CA, Ezugwu AL, Eze SO, Anyawu C, et al. Extraction, partial purification and characterization of pectinases isolated from Aspergillus species cultured on mango (mangifera indica) peels. African Journal of Biotechnology, (2014); 13(24): 2445-2454.
- Embaby AM, Masoud AA, Marey HS, Shaban NZ, Ghonaim TM. Raw agro-industrial orange peel waste as a low cost effective inducer for alkaline polygalacturonase production from Bacillus licheniformis SHG10. SpringerPlus, (2014); 3(1): 1-13.
- Soriano M, Diaz P, Pastor FJ. Pectinolytic systems of two aerobic sporogenous bacterial strains with high activity on pectin. Current microbiology, (2005); 50(2): 114-118.
- Jahan N, Shahid F, Aman A, Mujahid TY, Qader SAU. Utilization of agro waste pectin for the production of industrially important polygalacturonase. Heliyon, (2017); 3(6): e00330.
- Ghani M, Ansari A, Aman A, Zohra RR, Siddiqui NN et al. Isolation and characterization of different strains of Bacillus licheniformis for the production of commercially significant enzymes. Pakistan Journal of Pharmaceutical Sciences, (2013); 26(4): 691-697.
- Afifi A, Fawzi E and Foaad M. Purification and characterization of a pectin lyase produced by Curvularia inaequalis NRRL 13884 on orange peels waste, solid state culture. Annals of microbiology, (2002); 52(3): 287-298.
- Atalla S M, Aliaa R and El-Gamal NG. Cell immobilization of hydrolytic enzymes on olive mill wastes by Trichoderma harzianum NRC 12 and its antifungal activities aganist soil born fungi. Der Pharma Chemica, (2015); 7(4): 294-302.
This work is licensed under a Creative Commons Attribution-Non Commercial 4.0 International License. To read the copy of this license please visit: https://creativecommons.org/licenses/by-nc/4.0